An Antipsychotic Drug Is Being Recalled Because It May Contain Epilepsy Medication Instead
Please be careful!
You may want to check your medicine cabinet because yet another recall from Health Canada could impact you.
In a news release on Saturday, October 9, the government agency said that Riva-Risperidone 0.25 mg tablets are being recalled due to a "packaging error."
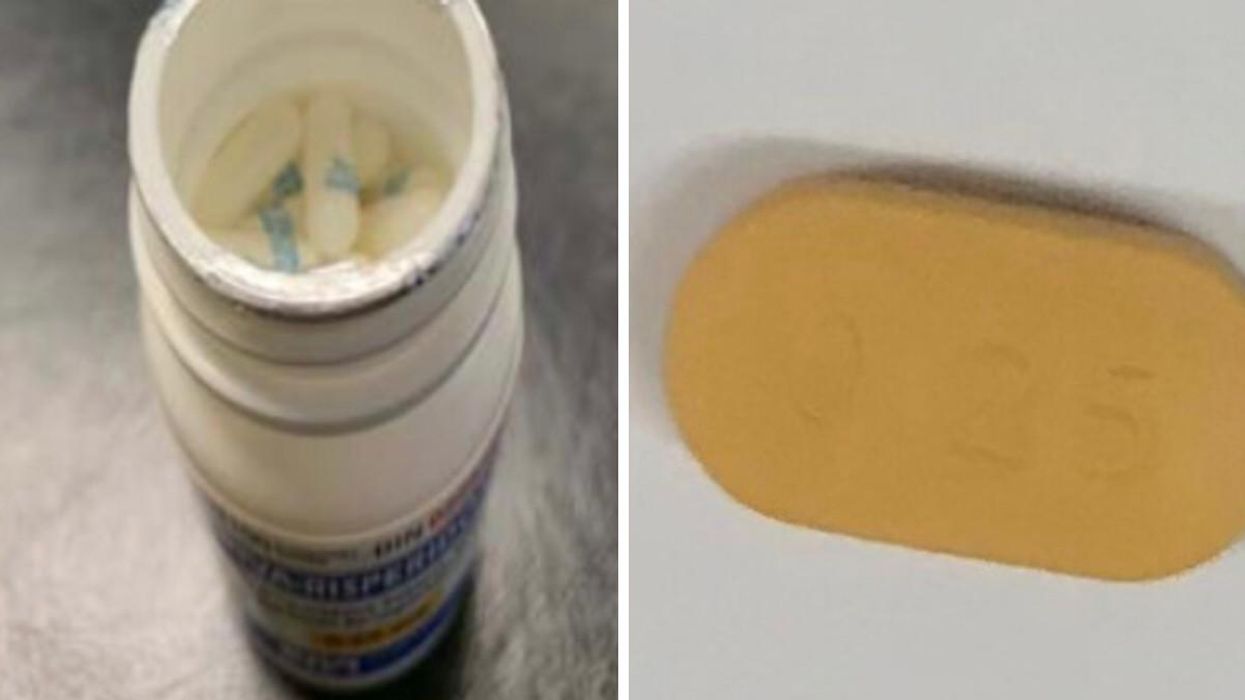
Apparently, some of the bottles may incorrectly contain only Riva-Gabapentin 100 mg capsules, which is a different prescription drug used for treating epileptic seizures.
According to Health Canada, Risperidone is a prescription drug used to treat psychotic disorders in adults, including schizophrenia, as well as bipolar disorder, and it can also be used for the short-term treatment of severe dementia related to Alzheimer's disease. However, Gabapentin is used for treating epileptic seizures.
"Pharmacists may not recognize the error and inadvertently repackage and dispense pill bottles that contain the wrong medication," the recall read.
As a result, Laboratoire Riva Inc. is recalling lot C9323 with an expiration date of March 31, 2022.
"Riva-Risperidone 0.25 mg tablets are yellowish-orange, oblong-shaped coated tablets, with '0.25' on one side and 'R' on the other side. Riva-Gabapentin 100 mg capsules are white hard gelatin capsules, with 'Gabapentin / 100 mg' printed on the capsule in blue ink," the recall added.
Health Canada is asking anyone who has a bottle that contains white capsules, or if you are unsure, to return it to your pharmacy as soon as possible. They said the pharmacist will check it and provide a replacement if needed.
Alternatively, anyone taking the medication who notices that their condition or symptoms are getting any worse, or is experiencing any side effects, is being asked to contact their health care provider immediately.